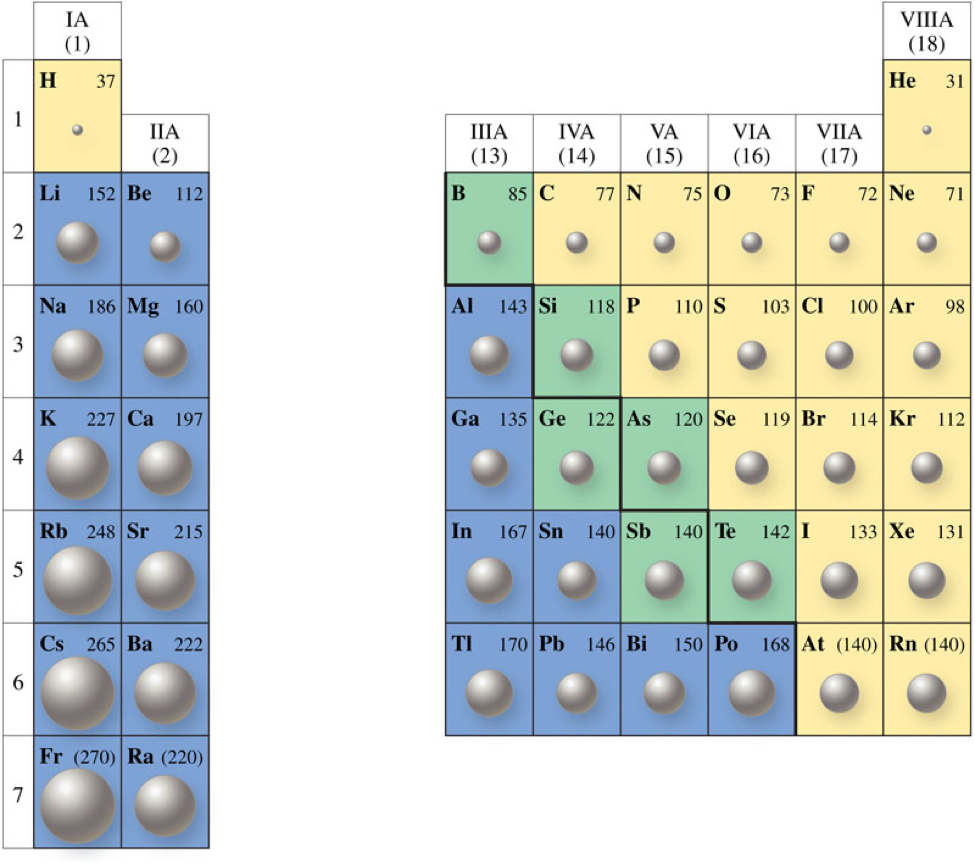
So we're going to say here, when we're looking at Ionic Radius, we're looking at ions and ions. The Ionic Radius increases as the number of electrons of that ion increases. Or let's say Ionic Radius equals the distance between ions, nucleus and its outer shell. Ionic Radius does not involve looking at the periodic table. Now Ionic Radius doesn't fit into this idea. 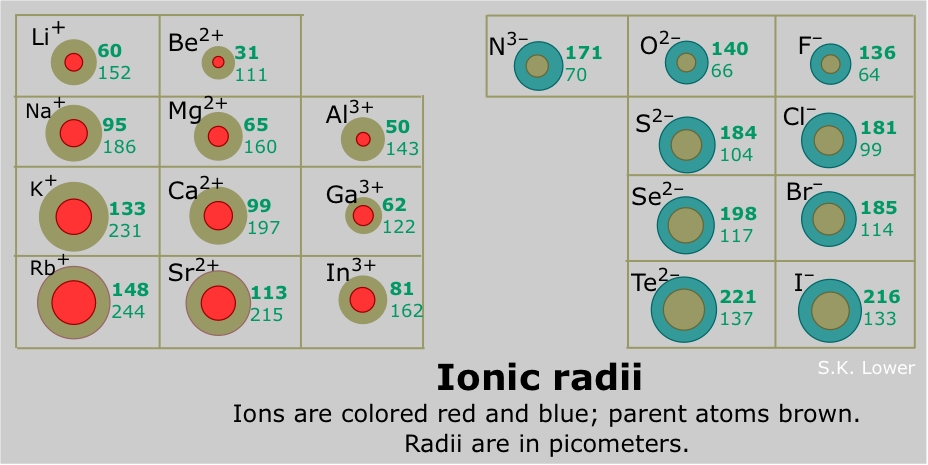
The periodic trends looking at the periodic table can tell you if they're increasing or decreasing in all our periodic trends were heading towards the top right corner and heading in that direction either increase or decrease that particular periodic trend. We conclude that the metallic radius is the distance between two atoms connected by a metallic connection.For a majority. 
However, because electrons don’t stay in one place, they orbit around it, the real measurement of the radius is a bit hazy. Its size is comparable to that of a metallic atom. Metallic radii, on the other hand, are the radii of metallic atoms. The atomic radius is the measurement of the distance between the nucleus and the electron in an atom. The covalent radius refers to the distance between nonmetallic components. The atomic radius of metals covalently linked together in a lattice is known as the covalent radius or metallic radius. For noble gas atoms, this is the most common radius reported. The “just barely touching” atomic radius is also known as the van der Waals radius, because the distance between the atoms is determined by the weak attraction of van der Waals forces. Atomic electron shells can overlap and overlap by varied distances depending on the circumstances. The distance between the nuclei of atoms and ions is affected by their arrangement or stacking. The atomic radius and ionic radius of an atom are both variable. The ionic radius of transition metals in a period, on the other hand, does not vary significantly from one atom to the next near the start of a series. These atoms lose their outermost electron, and in some cases, a complete electron shell is lost. The tendency is especially noticeable in metals that create cations. This is because the nucleus’ effective positive force increases, bringing electrons in even more tightly. The ionic and atomic radius decreases as the size of the atomic nucleus grows with greater atomic numbers travelling throughout a period. The atom’s overall size grows as a result of this.Ģ.The ionic radius reduces as you move from left to right across an element period (row). This is because as you progress down the periodic table, a new electron shell is added. In the periodic table, ionic and atomic radius follow the similar patterns:ġ.The ionic radius of an element group (column) rises as you move from top to bottom. X-ray crystallography or comparable techniques can be used to determine ionic radius. A common ionic radius ranges from 30 picometers (pm, or 0.3 Angstroms) to 200 pm (2 ). Ionic radius values are tough to come by and are often dependent on the method employed to quantify the ion’s size. An anion has an extra electron, which expands the electron cloud and may cause the ionic radius to exceed the atomic radius. Because an electron is removed, the remaining electrons are more firmly pushed in toward the nucleus, cations are typically smaller than neutral atoms. Because the boundary of an atom’s electron shell is somewhat hazy, ions are frequently represented as solid spheres fixed in a lattice.ĭepending on the electric charge of the ion, the ionic radius might be bigger or smaller than the atomic radius (radius of a neutral atom of an element). It is the half-distance between two ions that are barely touching. In a crystal lattice, the ionic radius (plural: ionic radii) is the measurement of an atom’s ion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
